|
In other words, there has always been, and always will be, exactly the same amount of energy in the universe. If such processes are present, repulsive forces must result which may lead eventually to. The first law of thermodynamics states that the total amount of energy in the universe is constant and conserved. but which, on the other hand, undergo catabolic processes. To appreciate the way energy flows into and out of biological systems, it is important to understand two of the physical laws that govern energy. For example, electrical energy, light energy, and heat energy are all different types of energy. In general, energy is defined as the ability to do work, or to create some kind of change. The laws of thermodynamics govern the transfer of energy in and among all systems in the universe. 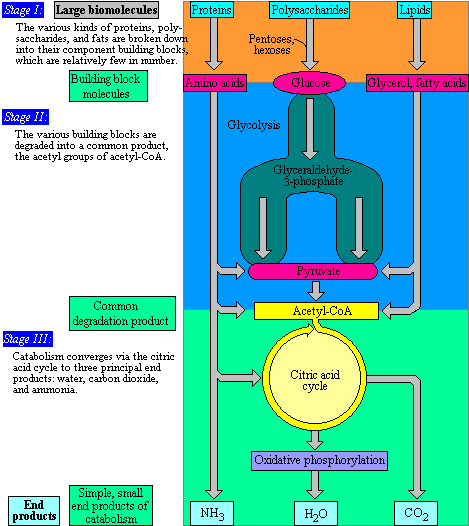
Like all things in the physical world, energy is subject to physical laws. Energy is exchanged between them and their surroundings as they use energy from the sun to perform photosynthesis or consume energy-storing molecules and release energy to the environment by doing work and releasing heat. One part of stage I of catabolism is the breakdown of food. In stage I, carbohydrates, fats, and proteins are broken down into their individual monomer units: carbohydrates into simple sugars, fats into fatty acids and glycerol, and proteins into amino acids. A closed system cannot exchange energy with its surroundings.īiological organisms are open systems. We can think of catabolism as occurring in three stages (Figure 26.5.1 26.5. The stovetop system is open because heat can be lost to the air. In an open system, energy can be exchanged with its surroundings. There are two types of systems: open and closed. Energy is transferred within the system (between the stove, pot, and water). 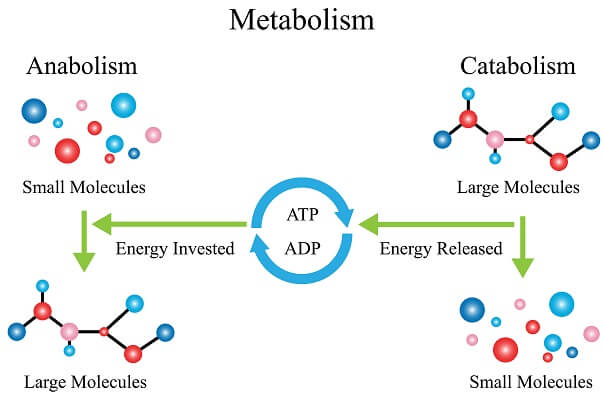
For instance, when heating a pot of water on the stove, the system includes the stove, the pot, and the water. The matter relevant to a particular case of energy transfer is called a system, and everything outside of that matter is called the surroundings. Thermodynamics refers to the study of energy and energy transfer involving physical matter. Both types of pathways are required for maintaining the cell’s energy balance. Anabolic pathways are those that require energy to synthesize larger molecules. We will now look at these three pathways.\): Catabolic pathways are those that generate energy by breaking down larger molecules. Anaerobic exergonic pathways do not require oxygen and include anaerobic respiration and fermentation. Aerobic respiration is an exergonic pathway that requires molecular oxygen (O 2). 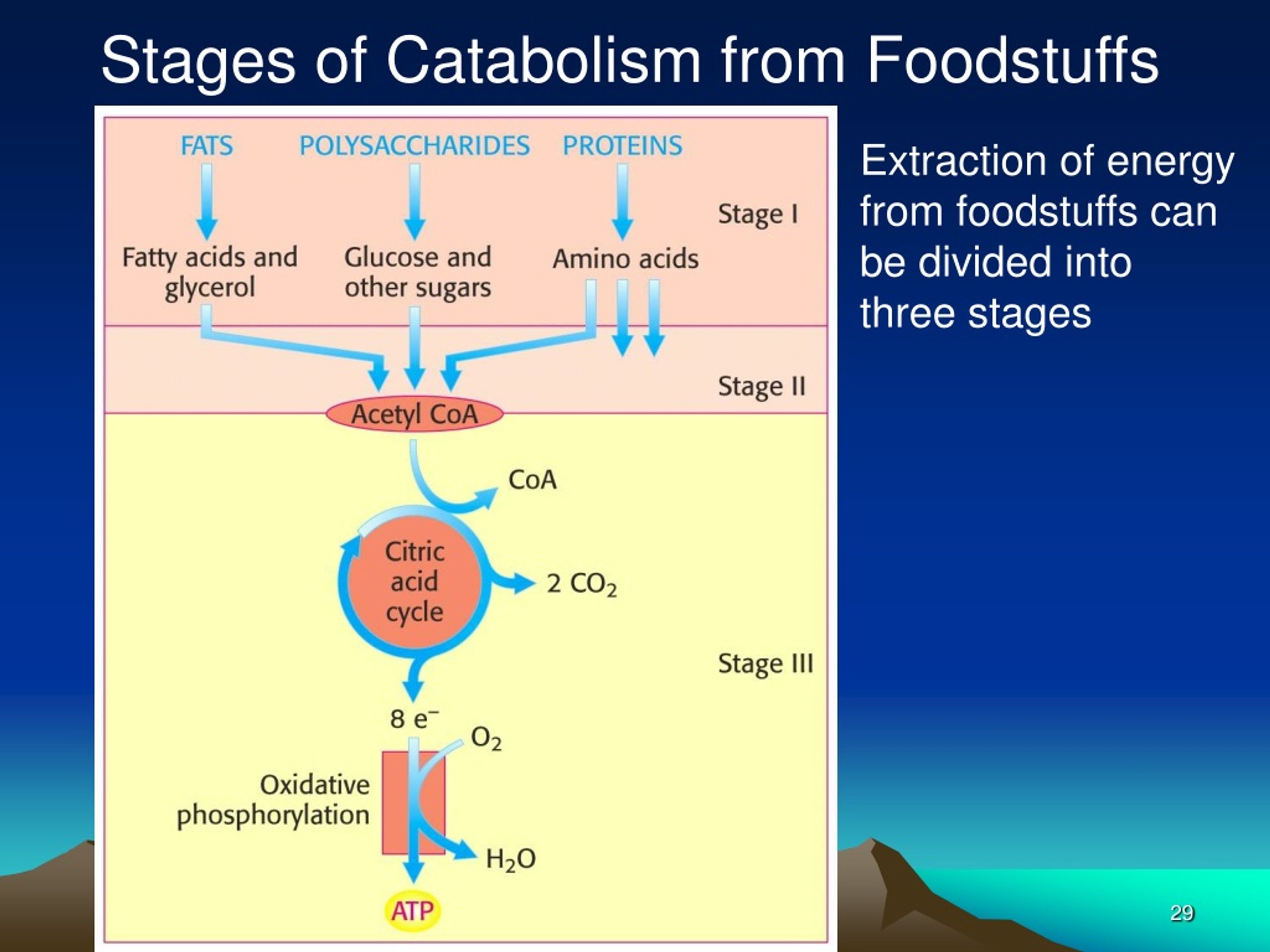
Depending on the organism, cellular respiration can be aerobic, anaerobic, or both. In this section we will concentrate primarily on harvesting energy and converting it to energy stored in ATP through the process of cellular respiration, but we will also look at some of the key precursor metabolites that are produced during this process.Ĭellular respiration is the process cells use to convert the energy in the chemical bonds of nutrients to ATP energy. 
Precursor metabolites are intermediate molecules in catabolic and anabolic pathways that can be either oxidized to generate ATP or can be used to synthesize macromolecular subunits such as amino acids, lipids, and nucleotides. Another factor that links catabolic and anabolic pathways is the generation of precursor metabolites. As can be seen, these two metabolic processes are closely linked. Anabolism is the endergonic process that uses the energy stored in ATP to synthesize the building blocks of the macromolecules that make up the cell. Living organisms are unique in that they can extract energy from their environments and use it to carry out activities such as movement, growth. Name one aerobic and two anaerobic forms of cellular respiration.Īs mentioned previously, to grow, function, and reproduce, cells must synthesize new cellular components such as cell walls, cell membranes, nucleic acids, ribosomes, proteins, flagella, etc., and harvest energy and convert it into a form that is usable to do cellular work.Ĭatabolism refers to the exergonic process by which energy released by the breakdown of organic compounds such as glucose can be used to synthesize ATP, the form of energy required to do cellular work. metabolism, the sum of the chemical reactions that take place within each cell of a living organism and that provide energy for vital processes and for synthesizing new organic material.Define precursor metabolites and state their functions in metabolism.\)ĭefine catabolism and anabolism and state which is exergonic and which is endergonic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
